Kaomei Guan Group
Application of patient-specific induced pluripotent stem cells in disease modelling

© Simon Lämmle
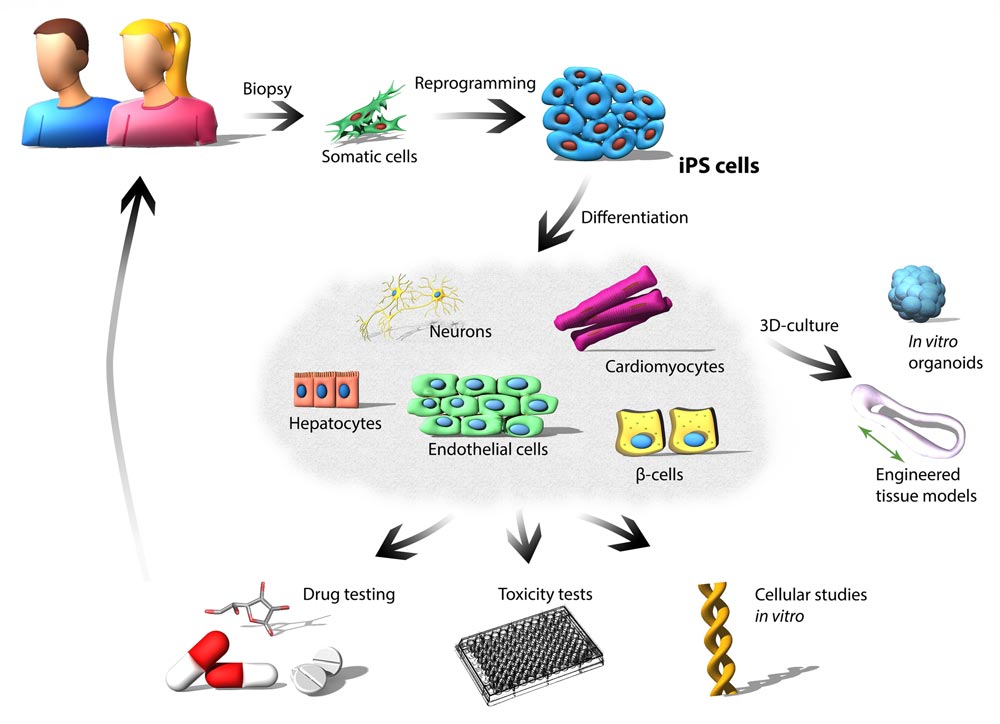
The research of our group is focused on the application of patient-specific induced pluripotent stem cells (iPS cells) for a deep understanding of disease pathogenic mechanisms, and identification of therapeutic targets for developing new treatment strategies and medications. Patient-specific iPS cells can be generated from somatic cells (e.g., skin cells, blood cells) by overexpressing specific transcription factors to reprogram cells to pluripotency. By this way, the cell gains two properties similar to embryonic stem cells. One is the self-renewal and the other is the differentiation potential (pluripotency). The self-renewal is a special form of unlimited proliferation to produce daughter cells that maintain the same properties of the parental stem cell. The differentiation potential allows the cells differentiating into specialized body cells (for example, heart cells, β-cells, nerve cells, hepatocytes, and muscle cells). Importantly, somatic cells derived from patient-specific iPS cells possess the same genetic background as the patient and display many of the characteristics of normal in vivo differentiated cells, including molecular, structural, and functional properties.
Genome-wide association studies indicate that many human diseases can be caused by monogenic defects in genes encoding structural or regulatory proteins. One focus of our work is the application of iPS cells as an in vitro disease model in the field of cardiovascular research and diabetes, in particular for the diseases with the early onset, to study the underlying molecular and pathophysiological mechanisms. We have established various iPS cell lines from patients who suffer from genetically induced diseases, including arrhythmias, hypertrophic or dilated cardiomyopathy. To analyze the diversity of the particular disease, we are using the CRISPR/Cas9 technology to edit the mutated genes involved in the patient-specific iPS cells, and thereby to “correct” them. Restoring the normal phenotype by correcting the target gene confirms that the mutation in the patient is the cause of the disease. Once we observe this, we can investigate how the malfunction of individual genes affects the development of the particular disease and identify therapeutic targets. Moreover, human iPS cell-based cell culture and in vitro generated organ-like tissues (organoids) are a promising approach to fill the gap between animal models and clinical trials in drug development.
We are focusing on the effect of experimental substances (for example, anti-arrhythmic drugs) – with specific versus multi-targeted effectiveness – on the properties of diseased cells, particularly, the extent that they lead to improved function. Patient-specific iPS cell-based models are particularly valuable and allow us investigating all pathophysiologic relevance when the disease affects multiple organs. Another key issue of the potential drugs during development is their toxicity. Cardiomyocytes and hepatocytes derived from human iPS cells represent a unique and predictive model to investigate the potential cardiotoxic and hepatotoxic effects of new pharmacological agents during the early development of drugs. For this purpose, we establish effective assays for drug toxicity tests by using the somatic cells differentiated from iPS cells.
Future Projects and Goals
- iPS-cell-based disease modelling for studying arrhythmia, hypertrophic cardiomyopathy or diabetes
- cGMP-cAMP crosstalk in iPS cell-derived cardiomyocytes
- the function of pituitary stem cells in states of perturbed STAT3-Ser/Hes3 Signaling using the human iPS cell system
- Establishment of human organoid cultures from patient-specific iPS cells (for example, cardiovascular, intestinal, cerebral, liver organoids and etc.)
Methodological and Technical Expertise
- Human and mouse induced pluripotent stem cells
- Cardiac differentiation
- Engineered heart tissues
- Disease modelling and drug testing
- CRISPR/Cas9 system