Anna Taubenberger Group
Oncomechanics

Current Research
Cancer cells and their surrounding tissue are characterised by altered mechanical properties. So far, it is not well understood how precisely these mechanical changes affect the cells’ metastatic potential in vivo. We address this question by mechanically characterising tumour samples to instruct physiologically relevant 3D in vitro models. These are then in combination with in vivo models employed to systematically study the role of cell and microenvironment stiffness on tumour spheroid growth, invasion, drug resistance, and metastasis. We aim at elucidating mechanisms by which cells sense their mechanical environment in 3D and how this can be potentially exploited for new therapeutic approaches.
Previous Research
Our previous work has shown that growth in stiff microenvironments causes compressive stress on breast cancer spheroids, which concomitantly alters their cytoskeleton organisation, increases constituent cell stiffness and slows down cell cycle progression. This contributes to a better understanding of how tumor cells adapt their proliferation under compressive stress in confining microenvironments. To characterise cell mechanical properties from single cells to complex tissues, a toolbox of atomic force microscopy, Brillouin microscopy and RT-DC is used.
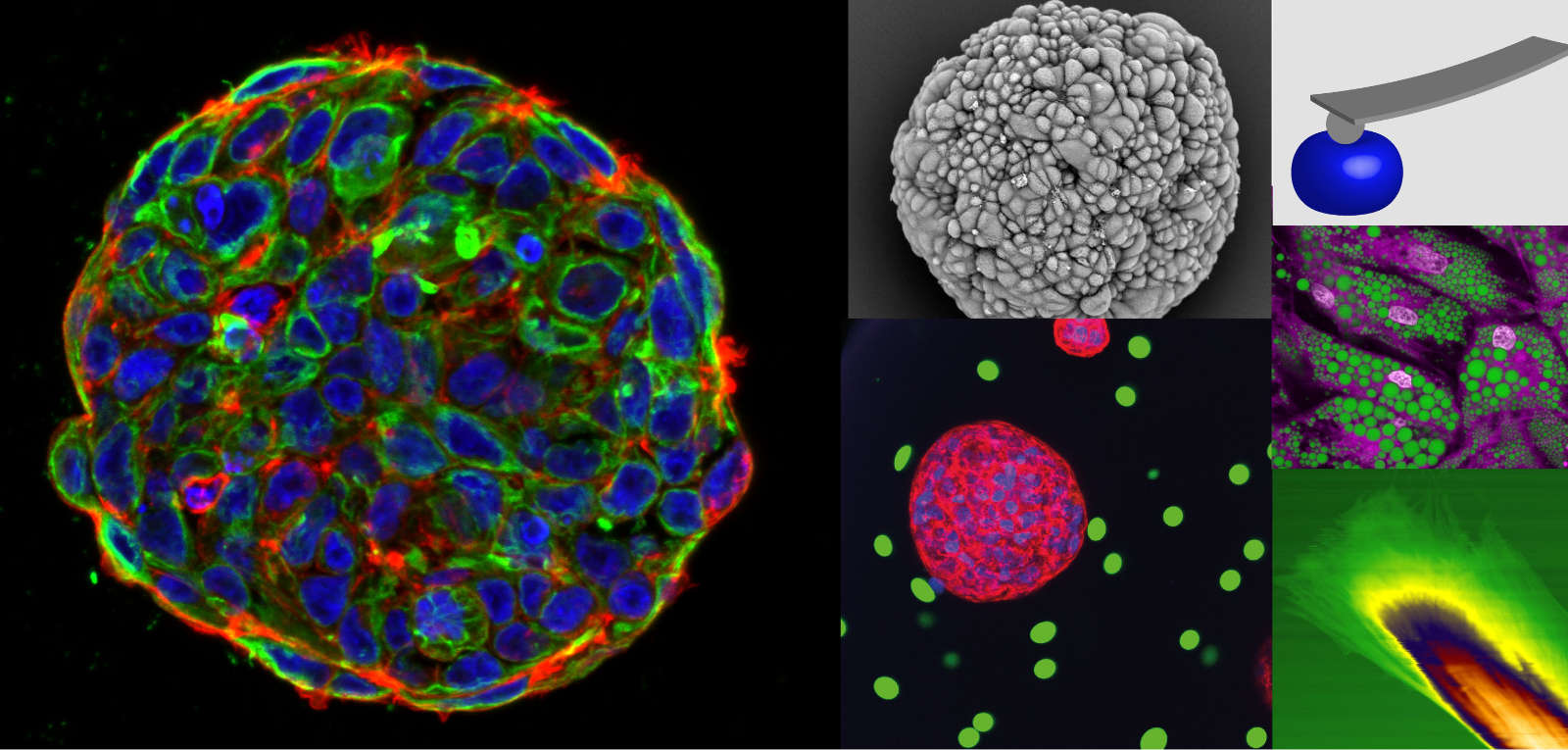
Future Projects and Goals
- Investigate the link between microenvironment mechanics and spheroid growth, mechanics, and invasion using bioengineered in vitro models.
- Test the functional role of cell mechanics in cancer cell invasion and metastasis using in vitro and in vivo models.
- Unravel mechanisms of how cells sense the mechanical properties of their 3D microenvironment.
- Identify clinical markers for individualized breast cancer therapy.
Methodological and Technical Expertise
- Atomic force microscopy (AFM)
- Real-time Deformability Cytometry
- 3D cultures (tumor spheroids, hydrogels)